General name
Desogestrel
ethynyl estradiol oral contraceptives do not prevent HIV infection (AIDS) and other sexually transmitted diseases (eg, syphilis, genital herpes, gonorrhea, chlamydia infections, condyloma acuminata, trichomoniasis vaginalis, hepatitis B, etc.) Fully explain to the recipient that the use of condoms is effective in preventing these infections.
If necessary, consider conducting a sexually transmitted disease test.
Contraindications (do not administer to the following patients)
1. Women with a predisposition to hypersensitivity to the ingredients of this drug
2. Estrogen-dependent malignant tumors (eg breast cancer, endometrial cancer), cervical cancer and patients suspected of having it [promotes tumor exacerbation or manifestation] Sometimes. ]
3. Patients with abnormal genital bleeding with unconfirmed diagnosis [Suspected genital cancer. If the bleeding is due to genital cancer, it may promote the exacerbation or manifestation of the cancer. ]
4. Patients with thrombophlebitis, pulmonary embolism, cerebrovascular accident, coronary artery disease or a history of it [Blood coagulation ability may be enhanced and these symptoms may be exacerbated. ]
5. Smokers aged 35 and over 15 cigarettes a day [It has been reported that cardiovascular disorders such as myocardial infarction are more likely to occur. ]
6. Patients with migraine with aura (scintillating scotch, star-shaped flash, etc.) [Reports that patients with migraine with aura are more likely to have cerebrovascular accidents (stroke, etc.) than patients without aura. There is. ]
7. Patients with valvular heart disease with pulmonary hypertension or atrial fibrillation, patients with valvular heart disease with a history of subacute bacterial endocarditis [Cardiovascular disorders such as thrombosis occur There are reports that it will be easier. ]
8. Diabetic patients with vascular lesions (diabetic nephropathy, diabetic retinopathy, etc.) [It has been reported that cardiovascular disorders such as thrombosis are more likely to occur. ]
9. Women with thrombophilia [It has been reported that cardiovascular disorders such as thrombosis are more likely to occur. ]
10. Patients with antiphospholipid antibody syndrome [It has been reported that cardiovascular disorders such as thrombosis are more likely to occur. ]
11. Patients within 4 weeks before surgery, within 2 weeks after surgery, within 4 weeks after childbirth and in a long-term resting state [Blood coagulation ability may be enhanced and the risk of cardiovascular side effects may increase. ]
12. Patients with severe liver damage
13. Patients with liver tumor [Symptoms may worsen. ]
14. Patients with dyslipidemia [It has been reported that cardiovascular disorders such as thrombosis are more likely to occur. In addition, symptoms may be exacerbated because it may affect lipid metabolism. ]
15. Patients with hypertension (excluding patients with mild hypertension) [It has been reported that cardiovascular disorders such as thrombosis are more likely to occur. In addition, the symptoms may worsen. ]
16. Patients with otosclerosis [Symptoms may worsen. ]
17. Patients with a history of jaundice, persistent pruritus or herpes during pregnancy [Symptoms may recur. ]
18. Pregnant or potentially pregnant women
19. Lactating women
20. Women who may not have completed bone growth [May cause premature closure of the epiphysis. 21. Patients receiving ombitasvir hydrate /
paritaprevir hydrate / ritonavir combination drug
Efficacy or effect
contraception
Precautions related to efficacy or effect
- Consider administration of this drug when it is considered inappropriate to administer other oral contraceptives. There are reports suggesting that it increases the relative risk of venous thrombosis compared to oral contraceptives such as levonorgestrel.
- It has been reported that the failure rate in general use including forgetting to take the oral contraceptive for one year is 9%.
Usage and dosage
- <21 tablet formulation>
Oral administration of 1 tablet daily at a fixed time for a total of 21 consecutive days, followed by a 7-day rest period. Repeatedly administer during the contraceptive period in a similar manner.
- <28 tablets>
One tablet daily is orally administered at a fixed time for 21 consecutive days, followed by green tablets for 7 consecutive days, for a total of 28 consecutive days. From the next cycle onward, administration will be continued from white tablets regardless of the presence or absence of withdrawal bleeding, and administration will be continued for 28 days. Therefore, no drug holiday will be taken from the start of administration in the first cycle. Menstruation (withdrawal bleeding) usually occurs while taking green tablets.
Precautions related to usage and dosage
- Take at a fixed time every day.
- Start date
When taking an oral contraceptive for the first time, start taking it on the first day of menstruation. If you start taking the medicine later than the first day of menstruation, you may become pregnant, so use other contraceptive methods for the first week of taking the medicine. - When administering this drug, give sufficient guidance on how to take it so that you do not forget to take it. In the unlikely event that you miss a dose (excluding the 28 green tablets), if you notice it by the next day, take the missed tablet immediately and have the tablet for that day taken as usual. If you miss a dose for 2 consecutive days or more, stop taking the drug, wait for the next menstruation, and resume administration.
If you miss a dose, you are more likely to become pregnant, so use another contraceptive method for that cycle. - When switching from other oral contraceptives to this drug
- When switching from 21-tablet type oral contraceptives Take
all the drugs you were taking before, and after a 7-day rest period, start taking this drug continuously. If you are late in taking the drug, you may be pregnant. - When switching from 28-tablet type oral contraceptives
After taking all the drugs that were taken before, start taking this drug continuously. If you are late in taking the drug, you may be pregnant.
- When switching from 21-tablet type oral contraceptives Take
Important basic notes
- Thrombosis may occur regardless of the presence or absence of risk factors such as age, smoking, obesity, and family history. If any of the following symptoms occur, administration should be discontinued immediately and appropriate. Performing the treatment.
- Main symptoms of thrombosis requiring emergency response
- Sudden pain / swelling of lower limbs, sudden shortness of breath, chest pain, severe headache, weakness / paralysis of limbs, dysarthria, acute visual impairment, etc.
- Main symptoms of thrombosis requiring emergency response
- Instruct people taking this drug to stop taking this drug immediately and consult an emergency medical institution if such symptoms occur.
- If symptoms of suspected thrombosis occur while taking this drug, take appropriate measures such as discontinuing administration.
- Symptoms of suspected thrombosis
- Lower limb pain / swelling / numbness / redness / heat sensation, headache, nausea / vomiting, etc.
- Symptoms of suspected thrombosis
- If there is an increased risk of thrombosis (immobility, marked increase in blood pressure, dehydration, etc.), appropriate measures such as discontinuation of administration should be taken.
- The following should be explained to the users of this drug at the start and continuation of administration.
- Thrombosis can have a life-threatening course.
- If symptoms of suspected thrombosis appear, or if the risk of thrombosis increases, stop taking the drug immediately and consult a doctor even if the symptoms / conditions are mild.
- If you suspect thrombosis and visit another medical institution, notify your doctor of the use of this drug so that you can receive a medical examination with this drug in mind.
- If it is unavoidable that surgery is necessary while taking this drug, give due consideration to the prevention of thrombosis.
- It has been reported that age and smoking volume increase the risk of serious cardiovascular side effects. Therefore, instruct people taking this drug to quit smoking.
- Before administration of this drug, it is necessary to investigate the medical history of the user and to have a medical examination. This examination includes blood pressure measurement, breast / abdominal examination and clinical examination. In addition, a medical examination should be performed every 6 months during administration.
- Before and during administration of this drug, the pelvic organs, mainly the uterus and ovaries, should be examined at least once a year. Consider performing cervical cytopathology once a year.
- For breast cancer testing, instruct the recipient to perform a self-examination.
- If vaginal bleeding develops while taking the drug, it usually disappears during continuous administration, but if it persists for a long period of time, it should be administered after confirming that it is not due to a malignant disease by examination such as vaginal cytopathology. ..
Composition / properties
composition
Maveron 21
| Common to Maveron 21/28 | ||
| Active ingredient | Desogestrel 0.15 mg in 1 tablet | |
| 1 tablet Chinese Pharmacopoeia ethinyl estradiol 0.03 mg |
||
| Additive | Potato starch, povidone, stearic acid, light anhydrous silicic acid, tocopherol, lactose hydrate, hypromellose, macrogol 400, titanium oxide, talc | |
Maveron 28
| Common to Maveron 21/28 | ||
| Active ingredient | Desogestrel 0.15 mg in 1 tablet | |
| 1 tablet Chinese Pharmacopoeia ethinyl estradiol 0.03 mg |
||
| Additive | Potato starch, povidone, stearic acid, light anhydrous silicic acid, tocopherol, lactose hydrate, hypromellose, macrogol 400, titanium oxide, talc | |
| Maveron 28 only | ||
| Active ingredient | Does not contain desogestrel in 1 tablet | |
| 1 tablet does not contain ethinyl estradiol |
||
| Additive | Corn starch, magnesium stearate, lactose hydrate, hypromellose, macrogol 400, titanium oxide, talc, yellow iron sesquioxide, edible blue No. 2 aluminum lake | |
Properties of the drug
Maveron 21
| Common to Maveron 21/28 | ||
| Dosage form | Film coated lock | |
| Tone | White | |
| External shape | surface |  |
| Back side |  |
|
| side | ||
| size | diameter | 5.0mm |
| thickness | 2.9mm | |
| mass | 66mg | |
| Identifying code | TR5 ☆ ORGANON |
|
| Number of locks | 21 tablets | |
Maveron 28
| Common to Maveron 21/28 | ||
| Dosage form | Film coated lock | |
| Tone | White | |
| External shape | surface |  |
| Back side |  |
|
| side | ||
| size | diameter | 5.0mm |
| thickness | 2.9mm | |
| mass | 66mg | |
| Identifying code | TR5 ☆ ORGANON |
|
| Number of locks | 21 tablets | |
| Maveron 28 only | ||
| Dosage form | Film-coated lock [placebo lock] | |
| Tone | green | |
| External shape | surface | 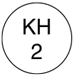 |
| Back side |  |
|
| side | ||
| size | diameter | 5.0mm |
| thickness | 2.8mm | |
| mass | 65mg | |
| Identifying code | KH2 ☆ ORGANON |
|
| Number of locks | 7 tablets | |
Contraindications for concomitant use (do not use together)
| Drug name, etc. | Clinical symptoms / measures | Mechanism / risk factors |
Ombitasvir hydrate / paritaprevir hydrate / ritonavir combination drug
|
Elevated ALT is frequently observed in patients who are concomitant with oral contraceptives containing ethinyl estradiol. Administration of this drug can be resumed approximately 2 weeks after the end of treatment with ombitasvir hydrate, paritaprevir hydrate, and ritonavir combination drug. |
Unknown mechanism |
Caution for combined use (Be careful about combined use)
| Drug name, etc. | Clinical symptoms / measures | Mechanism / risk factors |
Corticosteroids
Tricyclic antidepressant
Selegiline Hydrochloride |
The effects of these agents may be enhanced. | This drug is thought to suppress the metabolism of these drugs. |
| Rifampicin barbituric acid-based preparation
Hydantoin-based preparation
Carbamazepine |
The effect of this drug may be diminished and the incidence of genital bleeding may increase. | These drugs are thought to induce drug-metabolizing enzymes and promote the metabolism of this drug. |
Tetracycline antibiotics
Penicillin antibiotics
|
The effect of this drug may be diminished and the incidence of genital bleeding may increase. | It is considered that these drugs change the intestinal bacterial flora and suppress the reabsorption of this drug by enterohepatic circulation. |
| Terbinafine hydrochloride | It has been reported that menstrual disorders occurred when used in combination with a combination drug of progesterone and estrogen. | Unknown mechanism |
Gn-RH derivative
|
It may diminish the action of these agents. | Since these drugs show their efficacy by reducing the secretion of sex hormones, it is possible that the effects of these drugs may be diminished by administration of this drug, which is a sex hormone. |
Hypoglycemic agent
|
The action of hypoglycemic agents may be diminished. Carefully observe the blood glucose level and other conditions of the patient, and be careful to adjust the dose of hypoglycemic agent. | This drug is thought to reduce glucose tolerance and diminish the action of hypoglycemic agents. |
| Lamotrigine | Since it has been reported that the blood concentration of lamotrigine decreased when used in combination with an oral contraceptive, the dose adjustment of lamotrigine should be considered when starting or discontinuing the administration of this drug during the maintenance dose of lamotrigine. | Glucuronic acid conjugation of lamotrigine in the liver is promoted. |
| Morphine salicylic acid |
Blood levels of these drugs may decrease. | This drug is thought to promote glucuronidation of these drugs. |
| HIV Infectious Disease Treatment HIV Protease Inhibitor
Non-nucleoside reverse transcriptase inhibitor
|
The action of this drug may be diminished. | The AUC of ethinyl estradiol is reduced. |
|
The action of this drug may be diminished. | The blood concentration of etonogestrel, the active metabolite of desogestrel, decreases. |
|
The blood concentration of this drug may increase. | Etravirine is thought to inhibit the metabolic enzyme (CYP2C9) of this drug. |
HCV infection remedy
|
The action of this drug may be diminished. | The AUC of ethinyl estradiol is reduced. |
| Fluconazole Itraconazole |
The blood concentration of this drug may increase. | It is thought to inhibit the metabolic enzyme (CYP3A4) of this drug. |
| Voriconazole | The blood concentration of this drug may increase. Blood levels of voriconazole may increase. | Voriconazole is thought to inhibit the metabolic enzyme (CYP3A4) of this drug. This drug is thought to inhibit the voriconazole metabolizing enzyme (CYP2C19). |
| Acetaminophen | The blood concentration of this drug may increase. Blood levels of acetaminophen may decrease. | Acetaminophen is thought to inhibit the sulfate conjugate of ethinyl estradiol. This drug is thought to promote glucuronidation of acetaminophen in the liver. |
| Foods containing St. John's Wort | Since the effect of this drug may be diminished and the incidence of genital bleeding may increase, care should be taken not to ingest foods containing St. John's wort when administering this drug. | This food is thought to induce drug-metabolizing enzymes and promote metabolism of this drug. |
Side effects
The following side effects may occur, so observe carefully and take appropriate measures such as discontinuing administration if any abnormalities are observed.
Serious side effects
Thrombosis (limbs, lungs, heart, brain, retina, etc.) (incidence unknown)
If symptoms such as sudden pain / swelling of lower limbs, sudden shortness of breath, chest pain, severe headache, weakness / paralysis of limbs, dysarthria, acute visual impairment, etc. appear, administration should be discontinued immediately and appropriate measures should be taken. ..
Other side effects
| 5% or more | 0.1-5 % | Frequency unknown | |
| Hypersensitivity | rash | ||
| eye | Visual impairment | Visual impairment due to impaired retinal blood flow | |
| liver | Liver dysfunction, AST elevation, ALT elevation | jaundice | |
| metabolism | Edema and weight gain due to retention of sodium and fluids | ||
| Reproductive system | Vaginal bleeding (breakthrough bleeding, petechiae), bleeding, menorrhagia, dyspareunia, dyspareunia, diminished ribido | ||
| breast | Breast pain | Breast engorgement (feeling) | Milk leak |
| Cardiovascular | Extra systoles, increased blood pressure, palpitation | ||
| Digestive organ | nausea | Vomiting, diarrhea, abdominal pain, constipation, loss of appetite, heartburn, bloating | |
| Respiratory | Sore throat, cough | ||
| Psycho-nervous system | headache | Dizziness, drowsiness | Depression, irritability, migraine |
| Skin | Acne, eczema, itching, pigmentation, erythema | Hair loss | |
| Musculoskeletal | Back pain, leg pain, stiff shoulders, stiff fingers | ||
| others | Fatigue, dry mouth, facial edema, chest pain, leukopenia, elevated aldosterone |
Note) Be careful not to expose yourself to sunlight for a long time.
Impact on laboratory test results
Increases in serum proteins (corticoid-binding globulin, thyroxine-binding globulin, etc.) due to the action of the contained ethinyl estradiol may increase total cortisol, total T 3 , and total T 4 . Moreover, it is said that these free forms do not change. Be careful when determining these test values.
Precautions for application
Precautions when delivering drugs
Instruct them to take the medicine in the PTP package from the PTP sheet and take it. Accidental ingestion of the PTP sheet may cause a hard sharp corner to pierce the esophageal mucosa and further cause perforation, resulting in serious complications such as mediastinitis.
Other notes
Information based on clinical use
- As a result of epidemiological studies in foreign countries, it is reported that the risk of venous thrombosis is 3.25 to 4.0 times higher in women who take oral contraceptives than in women who do not. It has also been reported that the risk of venous thrombosis is highest during the first year of taking oral contraceptives. In addition, as a result of a large-scale post-marketing surveillance in a foreign country, not only when the oral contraceptive was first started, but also when the drug was resumed after a discontinuation of 4 weeks or more, or another oral contraceptive after a discontinuation of 4 weeks or more. It has been reported that the risk of venous thrombosis also increases when switching to, and the risk is particularly high for 3 months after the start of administration.
In addition, a report suggesting that oral contraceptives containing desogestrel from 1995 to 1996 increase the relative risk of venous thrombosis compared to oral contraceptives such as levonorgestrel (oral contraceptives such as levonorgestrel). There is 1 in 10,000 patients with venous thrombosis per year, compared to 2 with oral contraceptives containing desogestrel). By the way, it is said that the incidence of venous thrombosis due to pregnancy is 6 per 10,000 people in one year. - As a result of epidemiological studies in foreign countries, it has been reported that taking oral contraceptives increases the possibility of developing breast cancer and cervical cancer.
- It has been reported that benign liver tumors occur in 3.4 per 100,000 people when oral contraceptives are taken in foreign countries for 2 years or more. In addition, rupture of the tumor may cause intra-abdominal hemorrhage. On the other hand, the incidence of malignant liver tumor (liver cancer) is extremely low, less than 1 in 1 million.
- In foreign countries, it has been reported that taking oral contraceptives worsened systemic lupus erythematosus (SLE), anaphylaxis-like symptoms, hemolytic uremic syndrome (HUS), and angioedema.
- In foreign countries, it has been reported that contact lenses are not adjusted well due to changes in corneal thickness due to taking oral contraceptives, resulting in changes in visual acuity and visual field, and discomfort when wearing.
Pharmacokinetics
Blood concentration
When orally administered to healthy adult women, absorption from the gastrointestinal tract is rapid, and serum concentrations of the active metabolites 3-keto-desogestrel and ethinyl estradiol reach their maximum about 2 hours after administration. In addition, when repeated doses were administered, the serum concentrations of 3-keto-desogestrel and ethinyl estradiol reached a steady state on approximately 15 days, and the elimination phase half-lives after 21 days were 22.0 and 23.7 hours, respectively.
| Table 21 Pharmacokinetic parameters after continuous oral administration for 21 days | ||||
| AUC (pmol · hr / mL) |
t 1/2 (hr) |
C max (pmol / mL) |
T max (hr) |
|
| 3-Keto-Desogestrel | 97.2 ± 36.8 | 22.0 ± 9.6 | 12.12 ± 5.49 | 1.2 ± 0.6 |
| Ethinyl estradiol | 11.7 ± 3.3 | 23.7 ± 8.8 | 1.18 ± 0.32 | 1.1 ± 0.6 |
excretion
When 3 H-labeled desogestrel and ethinyl estradiol were orally administered to healthy adult women, about 48% was excreted in urine and about 35% in feces by the 8th day after administration (foreign data).
Clinical results
Efficacy and safety testing
Domestic clinical trials
In a clinical study conducted on 992 women who wished to contraceptive, one pregnancy was determined to be drug-induced, with a contraceptive effect of 99.9% and a Pearl index of 0.085 during the total dosing cycle (14,088 cycles). ..
Of the 1,011 women (14,378 cycles) who wished to contraceptive, side effects were reported in 258 (25.5%), mainly nausea in 119 (11.8%) and breast pain in 85 (8.4%). , Headache 59 cases (5.8%), irregular organ bleeding 24 cases (2.4%), vomiting 23 cases (2.3%), malaise 12 cases (1.2%), diarrhea 11 cases (1.1%), abdominal pain 10 cases (1.0%) )Met. Abnormal laboratory test values were reported in 2 cases with increased AST and ALT, 1 case with decreased white blood cell count, and 1 case with increased aldosterone, for a total of 4 cases.
others
Various contraceptive methods The failure rate (pregnancy rate) for one year after the start of use is as follows .
| Method | Ideal use (%) |
General use ) (%) |
| Oral contraceptive | 0.3 | 9 |
| Levonorgestrel release IUS | 0.2 0.2 | 0.2 0.2 |
| Copper IUD | 0.6 | 0.8 0.8 |
| condom | 2 | 18 18 |
| Rhythm method | 0.4-5 | twenty four |
| Female contraceptive surgery | 0.5 | 0.5 |
| Male contraceptive surgery | 0.10 | 0.15 |
| Without contraception | 85 | 85 |
| IUS : Intrauterine system IUD: Intrauterine device
Note 1) If you become pregnant even though you continue to use the selected contraceptive method correctly. Note 2) If you become pregnant despite using the selected contraceptive method (for oral contraceptives, the failure rate including forgetting to take) |
||
Pharmacology
Mechanism of action
This drug has an ovulation-suppressing effect as its main effect, and exerts a contraceptive effect by inhibiting implantation due to changes in the endometrium and inhibiting sperm passage due to changes in cervical mucus.
Ovulation inhibitory effect
By taking this drug continuously, the luteal and estrogen levels in the blood are kept constant. As a result, the physiological pattern of anterior pituitary gonadotropin secretion is inhibited, and ovulation is suppressed by reducing gonadotropin secretion.
Implantation inhibitory effect due to changes in the properties of the endometrium
Continuous administration of this drug changes the properties of the endometrium, resulting in inhibition of implantation of fertilized eggs.
Inhibition of sperm passage by changes in cervical mucus
Continuous administration of this drug causes changes in the properties and composition of cervical mucus, which impedes the passage of sperm into the uterine cavity.
Physicochemical knowledge about active ingredients
Desogestrel
General name
Desogestrel
Chemical name
(+)-17α-Ethynyl-18-methyl-11-methylene-4-estren-17-ol
Molecular formula
C 22 H 30 O
Molecular weight
310.47
Properties
It is a white crystalline powder with no odor.
Very soluble in acetone, dichloromethane or tetrahydrofuran, soluble in ethanol (99.5) or diethyl ether, slightly soluble in hexanes, practically insoluble in water.
Chemical structural formula
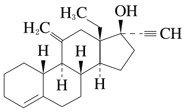
Melting point
110-112 ℃
Ethinyl estradiol
General name
Ethinylestradiol
Chemical name
19-Nor-17α-pregna-1,3,5 (10) -triene-20-yne-3,17-diol
Molecular formula
C 20 H 24 O 2
Molecular weight
296.40
Properties
White to slightly yellow crystals or crystalline powder with no odor.
It is freely soluble in pyridine or tetrahydrofuran, slightly soluble in ethanol (95) or diethyl ether, and practically insoluble in water. Soluble in sodium hydroxide test solution.
Chemical structural formula
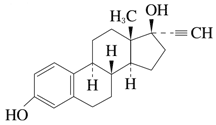
Melting point
180-186 ° C or 142-146 ° C
Handling precautions
Keep this drug out of the reach of children.
Packaging
- <Maveron 21>
210 tablets [21 tablets (PTP) x 10]
1,050 tablets [21 tablets (PTP) x 50]
- <Maveron 28>
280 tablets [28 tablets (PTP) x 10]
1,400 tablets [28 tablets (PTP) x 50]
Marvelon 21 / Marvelon 28
Creation or revision date
| * | Revised September 2021 (2nd edition) |
| Revised October 2019 (1st edition) |
Japanese standard product classification number
872549
Medicinal effect classification name
Oral contraceptive
Approval, etc.
Maveron 21
Brand name code
YJ code
254910CF1025
Brand name English notation
MARVELON 21 Tablets
Brand name Hiragana
Maberon
Approval number, etc.
Approval number
21100AMY00154000
Sales start date
April 2005
Saving method / validity period
Saving method
Store at room temperature
Validity period
3 years
Regulation classification
Prescription drugs
Note 1) Caution-Use according to the doctor's prescription
Maveron 28
Brand name code
YJ code
254910CF2021
Brand name English notation
MARVELON 28 Tablets
Brand name Hiragana
Maberon
Approval number, etc.
Approval number
21100AMY00155000
Sales start date
December 2006
Saving method / validity period
Saving method
Store at room temperature
Validity period
3 years
Regulation classification
Prescription drugs
Note 2) Caution-Use according to the doctor's prescription
Main literature
1) Masahiko Mizuno et al . Journal of the Japanese Society of Infertility . 1991; 36: 35-48.
2) Viinikka L, et al. Acta Endocrinol. 1980; 93: 375-9.
3) Masahiko Mizuno et al . Obstetrics and Gynecology . 1990; 57: 2507-32.
4) Shigeo Takagi et al . Basic and clinical . 1991; 25: 2807-16.
5) Masahiko Mizuno et al . Obstetrics and Gynecology . 1992; 59: 149-59.
6) Hatcher RA, et al. Contraceptive Technology. Twentieth Revised Edition. NewYork: Ardent Media; 2011
7) Cullberg G, et al. Acta Obst Gynecol Scand. 1982; Suppl. 111: 29-38.
8) Mall-Haefeli M, et al. Geburtshilfe Frauenheilkd. 1984; 44: 177-9.
9) Viinikka L, et al. Acta Endocrinol. 1976; 83: 429-38.
10) Safety References Tumor Matters
• Collaborative Group on Hormonal Factors in Breast Cancer : Lancet, 347 : 1713-1727, 1996
• WHO : Int J. Cancer, 55 : 228-236, 1993
• Ursin, G. et al . : Lancet, 344 : 1390-1394, 1994
• Ye, Z. et al . : Int J. Epidemiol., 24 : 19-26, 1995
• Thomas, D. B. et al . : Am. J. Epidemiol., 144 : 281-289, 1996
• Shigeo Ogawa et al .: Mandatory Obstetrics and Gynecology Revised 4th Edition , p.390, p.395-396, Nanedo, 1996 •
Edmondson, H.A. et al . :N. Engl. J. Med., 294 : 470-472, 1976
• Neuberger, J. et al . : Br. Med. J., 292 : 1355-1357, 1986
• Principles of Obstetrics and Gynecology, Gynecology , p. 618-623, Medical View , 1987
• Breast Cancer Screening (Japan vs. Cancer Association , Social Insurance Publishing , 1984 )
• Breast Cancer Mass Screening Guide (Breast Cancer Study Group , Shinohara Publishing , 1987 )
• Breast Cancer Diagnosis and Treatment (Pharmaceuticals ) Journal , 1995 )
• Becker, T.M. et al . : Int. J. Epidemiol., 23 : 913-922, 1994
• Parazzini, F. et al . : Br. J. Cancer, 77 : 838-841, 1998
• Zondervan, K.T. et al . : Br. J. Cancer, 73 : 1291-1297, 1996
• Nobutaka Nagai: Rinjo , 51 : 429-436, 1997
• Chie, WC. Et al . : Int. J. Cancer , 77 : 219-223, 1998
• Rooks, J.B. et al . : JAMA, 242 : 644-648, 1979
• Corfman, P.A . : Contraception, 37 : 433-455, 1988
• Mays, E.T. et al . : Semin. Liver Dis ., 4 : 147-157, 1984
• Forman, D. et al . : Br. Med. J., 292 : 1357-1361, 1986
• WHO :Int. J. Cancer, 43 : 254-259, 1989
• The Collaborative MILTS ( Multi-centre International Liver Tumor Study ): Contraception, 56 : 275-284, 1997
11) Safety References Cardiovascular
• WHO : Lancet, 346 : 1575-1582, 1995
• Spitzer, W.O. et al . : Br. Med. J., 312 : 83-88, 1996
• WHO : Lancet , 348 : 498-505, 1996
• WHO : Lancet, 348 : 505-510, 1996
• Petitti, D.B. et al . : N. Engl. J. Med., 335 : 8-15, 1996
• WHO : Lancet, 349 : 1202-1209, 1997
• Lewis, M.A. et al . : Contraception, 56 : 129-140, 1997
• WHO Special Program of Research: Br. J. Obstet. Gynaecol., 98 : 1117-1128, 1991
• Jespersen, J. et al . : Am. J. Obstet. Gynecol., 163 : 396-403, 1990
• Layde, P. M. et al . : Lancet, I : 541-546, 1981
• Stadel, B.V . : N. Engl. J. Med., 305 : 672-677, 1981
• Mutsumi Kazama: Biomedical Perspectives, 6 : 85-89, 1997
• Bloemenkamp, K.W.M. et al . : Lancet, 346 : 1593-1596, 1995
• Masao Maruyama: Thrombosis and Circulation , 4 : 21-23, 1996
• Junzo Nojima et al .: Clinical Pathology , 46 :1181-1187, 1998
• Robinson, G.E. et al . : Br. Med. J., 302 : 269-271, 1991
• Vessey, M.P. et al . : Br. Med. J., 3 : 123-126, 1970
• Nobutoshi Shinoki et al .: medicina, 33 : 1308-1310, 1996
• Gibbs, N.M . : Br. J. Surg., 45 : 209-236, 1957
• Kawasaki, T. et al . : Thrombosis Res., 79 : 147-151 , 1995
• Shigeo Takagi et al .: Sogo Clinic , 34 : 1115-1119, 1985
• Jain, AK. Et al . : Studies in Family Planning, 8 : 50-54, 1977
• "Committee Statement for Oral Contraceptives Containing Desogestrel or Gestodene ( CPMP ) " , CPMP / 374/96 , April 17 , 1996 • Nobuo Sakuragawa et al .: Clinical Thrombosis Hematology, p.231, Medical and Dental Publishing , 1994 • Yukiichi Wakisaka et al .: Pathophysiology , p.477, Nankodo , 1978 • Farley, T.M.M. et al . : Contraception, 57 : 211-230, 1998 • Heinemann, L.A. J. et al . : Br. Med. J., 315 : 1502-1504, 1997
12) Safety reference materials Matters concerning the impact on the next generation
• Yoshiko Yasuda et al .: History of medicine , 98 : 537-538, 1976
• Yoshiko Yasuda et al .: History of medicine , 99 : 611-612, 1976
• Takasugi, N . et al . : J. Natl. Cancer Inst., 33 : 855-865, 1964
• Takao Mori: History of Medicine , 95 : 599-602, 1975
• Huggins, G. R. et al . : Fertil. Steril., 54 : 559-573, 1990
• Corfman, P. A . : Contraception, 37 : 433-455, 1988
• Heinonen, O. P. et al . : N. Engl. J. Med., 296 : 67-70, 1977
• Harlap, S. et al . : Obstet. Gynecol., 55 : 447-452, 1980
• Savolainen, E. et al . : Am. J. Obstet. Gynecol., 140 : 521-524, 1981
• Ferencz, C . et al . : Teratology, 21 : 225-239, 1980
• Rothman, K. J. et al . : Am. J. Epidemiol., 109 : 433-439, 1979
• Kunijun Tsunemi et al .: Basic and Clinical , 24 : 4737- 4756, 1990
• Shigeo Harada et al .: Pharmacology and Treatment , 19 : S965-S999, 1991
• Kwarta, Jr. R. F. et al . : Applied Pharmacology , 42 : 327-340, 1991
• Akihiko Kageyama et al .: Nikkei Medical Bulletin, 36 : 284-299, 1991
• 20 -year history of birth defect investigation : Japan Maternal Protection Medical Association , 1993
• Congenital anomaly birth monitoring survey at Tokyo Metropolitan Hospital
• Li, D.K. et al . : Teratology, 51 : 30-36 , 1995
• Kim, M. R. et al . : Am. J. Obstet. Gynecol., 172 : 1042-1043, 1995
• Naotaka Ishizuka: Journal of the Endocrine Society of Japan , 38 : 443-449, 1962
• Carr, D. H. et al . : Can. Med. Assoc. J., 103 : 343-348, 1970
• Lauritsen, J. G . : Acta Obstet. Gynecol. Scand., 54 : 261-264, 1975
• Dhadial, R. K. et al . :Lancet, II : 20-21, 1970
• Boue, J. et al . : Teratology, 12 : 11-26, 1975
• Hide Matsunaga: Obstetrics and Gynecology Treatment , 32 : 88-93, 1976
• Harlap, S. et al . : Teratology, 31 : 381-387, 1985
• Depue, R. H. et al . : J. Natl. Cancer Inst., 71 : 1151-1155, 1983
• Henderson, B. E. et al . : Int. J. Cancer, 23 : 598-602, 1979
• Brown, L. M. et al . : Cancer Res., 46 : 4812-4816, 1986
• Moss, A. R. et al . : Am. J. Epidemiol., 124 :39-52, 1986
13) Safety References Pregnancy Function
• Woutersz, T.B . : J. Reprod. Med., 26 : 615-620, 1981
• Royal College of General Practitioners Oral Contraceptives and Health, p.71-77, Pitman Medical, London, 1974
• Mosher, W. D. et al . : Fertil. Steril., 56 : 192-193, 1991
• Chasan-Taber, L. et al . : Am. J. Epidemiol., 146 : 258-265, 1997
14) Safety References Others
• Ministry of Health, Labor and Welfare Drug Side Effect Information , No.28, 33-35, 1977
• Fisch, I.R. et al . : JAMA, 237 : 2499-2503, 1977
• Schwartman JA et al . : Rev. Bras . Oto-Rino-Laringol., 41 : 46-50, 1975
• Drill, V. A . : Am. J. Obstet. Gynecol., 119 : 165-174, 1974
• Orellana-Alcalde, J. M. et al . : Lancet, II : 1278-1280, 1966
• Koide, S. S. et al . : J. Reprod. Med., 15 : 214-224, 1975
• Nilsson, S . : Contraception, 17 : 131-139, 1978
• Masaaki Yoshimoto et al .: Hormones and Clinic , 44 : 597-605, 1996
• Chasan-Taber, L. et al . : Circulation, 94 : 483-489, 1996
• Godsland, IF et al . : J. Clin. Endocrinol. Metab., 74 : 64-70, 1992
• Ryoko Ninomiya et al .: Clinical Dermatology , 48 : 281-283, 1994
• Kenichi Furihata et al .: Latest Medicine , 35 : 2522-2527, 1980
• Harrison's Principles of Internal Medicine, Eighth Edition , p.176, p.1450, 1977
• Koji Okada: The ideal way of steroid therapy Obstetrics and Gynecology , p.171, Kinhodo , 1982
• Zimmerman, A.W . : Neurol. Clin. 4 :853-861, 1986
• Burckhardt, P . : Hormone Res., 6 : 321-328, 1975
• Crook, D. et al . : Am. J. Obstet. Gynecol., 158 : 1612-1620, 1988
• Frey, B. M. et al . : Eur. J. Clin. Pharmacol., 26 : 505-511, 1984
• Abernethy, D.R. et al . : Clin. Pharmacol. Ther., 35 : 792-797, 1984
• Selegiline hydrochloride Attachment
• Cyclosporin Attachment
• Reimers, D. et al . : Prax. Pneumol., 25 : 255-262, 1971
• Back, D. J. et al . : Contraception, 21 : 135-143, 1980
• Back, D. J. et al . : Br. J. Pharmacol., 69 : 441-452, 1980
• Back, D. J. et al . : Br. J. Clin. Pharmacol., 25 : 527-532, 1988
• Crawford, P . et al . : Br. J. Clin. Pharmacol., 30 : 892-896, 1990
• Back, DJ et al . : J. Steroid. Biochem., 16 : 407-413, 1982
• Terbinafin Hydrochloride Attachment
• Buserelin acetate Attachment
• Steel, J.M . : J. Fam. Plan. Doctors, 3 : 77-78, 1978
• Nerphinavir mesylate Attachment
• Ouellet, D. et al . : Br. J. Clin. Pharmacol., 46 :111-116, 1998
• Nevirapine package insert
• Yuko Higaki: Today's Treatment Guidelines , p713, Igaku-Shoin, 1999 •
Van Vange, N. et al . : Contraception, 41 : 345-352, 1990
• Minoru Noguchi: Nissan Women's Magazine , 40 : 14-20, 1988
• Schatz, D.L. et al . : Can. Med. Assoc. J., 99, 882-886, 1968
• Shuichi Imagama et al .: Clinical Ophthalmology , 41 : 751-753, 1987
• Beral, V. et al . : Br. Med. J., 318 : 96-100, 1999
• Mostad, S. B. et al . : Lancet, 350 : 922-927, 1997
• Royal College of General Practitioners: Oral Contraceptives and Health, p.31, Pitman Medical, London, 1974
• Bray, R.S . : Contraception, 13 : 417-425, 1976
• Keller, A. J. et al . : Obstet. Gynecol., 49 : 83-91, 1977
• Gerretsen, G. et al . : Contraception, 22 : 25-29, 1980
• Baker, D.A. et al . : Contraception, 32 : 377-382, 1985
• Bisset, L. R. et al . : Contraception, 38 : 573-578 , 1988
• Baker, D.A. et al . : Contraception, 39 : 119-124, 1989
• Scanlan, J.M. et al . :Psychoneuroendocrinology, 20 : 281-287, 1995
15) Effectiveness References Contraceptive Effects
• Hatcher, R.A. et al . : Contraceptive Technology : Twentieth Revised Edition. New York : Ardent Media , : p. 779-861, 2011
• Morris, S.E. et al . : Contraception, 20 : 61-69, 1979
• Chowdhury, V. et al . : Contraception, 22 : 241-247, 1980
• Sparrow, M.J . : N.Z. Med. J., 100 : 102-105, 1987
16) Reference materials on effectiveness Materials on abortion
• 1997 Maternal Protection Statistics Report ( Vital Statistics Division, Statistics Information Department, Minister of Health and Welfare)
• Mainichi Shimbun Population Problem Survey Committee ( 23rd National Family Planning Opinion Poll Report) • Determinants of
contraceptive use
• United Nations : Demographic Yearbook 1996 Edition . © 1998
17) Effectiveness References • Side
Benefits • The Cancer and Steroid Hormone Study of the Centers for Disease Control and the National Institute of Child Health and Human Development : N. Engl. J. Med., 316 : 650-655, 1987
• Narod, S.A. et al . : N. Engl. J. Med., 339 : 424-428, 1998
• The Centers for Disease Control Cancer and Steroid Hormone Study : JAMA, 249 : 1596-1599, 1983
• The Cancer and Steroid Hormone Study of the Centers for Disease Control and the National Institute of Child Health and Human Development : JAMA, 257 : 796-800, 1987
• Schlesselman, J.J . :Hum. Reprod., 12 : 1851-1863, 1997
• Royal College of General Practitioners : Oral Contraceptives and Health, p.22, p.61, Pitman Medical, London, 1974
• Connel, E.B. et al . : J. Reprod. Med., 29 : 513-523, 1984
• Lanes, S.F. et al . : Am. J. Obstet. Gynecol., 166 : 956-961, 1992
• Vessey, M. et al . : J. Biosoc. Sci ., 8 : 373-427, 1976
• Westrom, L . : Am. J. Obstet. Gynecol., 138 : 880-892, 1980
• Rubin, G. L. et al . : Am. J. Obstet. Gynecol., 144 : 630- 635, 1982
• Brinton, L. A. et al . : Am. J. Epidemiol., 113 : 203-214, 1981
Document request and contact information
Organon Co., Ltd. Customer Support Center
1-24-3 Minami Aoyama, Minato-ku, Tokyo
Toll free number 0120-095-213
Precautions for insurance benefits
This drug is not covered by insurance benefits (not listed in the drug price standard).
Manufacturers, etc.
Manufacturer and distributor
Organon Co., Ltd.
1-24-3 Minami Aoyama, Minato-ku, Tokyo

